This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Photocatalytic nonoxidative coupling of methane to ethylene over carbon-doped ZnO/Au catalysts
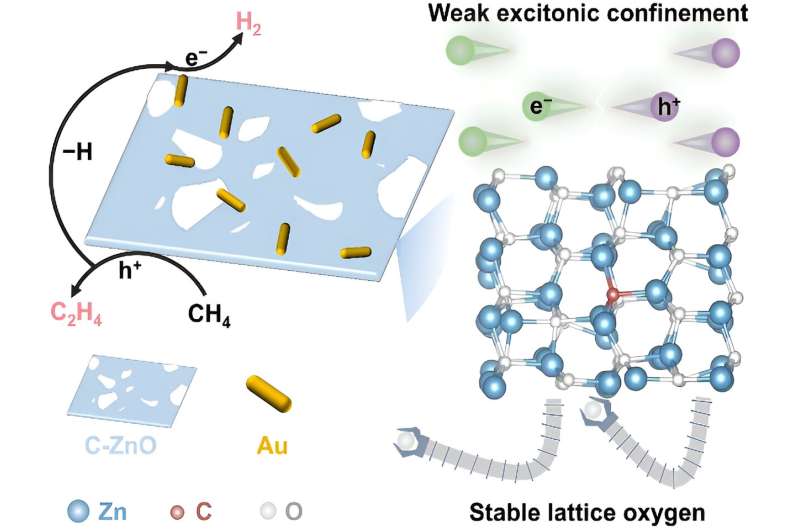
A study on the photocatalytic nonoxidative coupling of methane to ethylene over carbon-doped ZnO/Au catalysts was published by Prof. Wei Xiao (College of Chemistry and Molecular Sciences, Wuhan University) and Dr. Yuhao Peng (College of Chemistry and Molecular Sciences, Wuhan University).
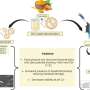
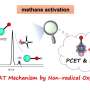
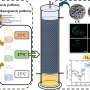
A photocatalytic conversion of methane to multi-carbon compounds remains a huge challenge due to its high dissociation energy of C−H bonds and sluggish charge carrier dynamics. Au-modified carbon-doped ZnO (C-ZnO/Au) photocatalyst is constructed by an interfacial modification-assisted self-assembly approach for photocatalytic conversion of methane.
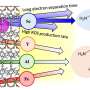
Benefitting from the presence of C-ZnO/Au interfaces, the catalyst not only reduces the excitonic binding energy to improve the photogenerated charge carrier separation but also enhances the stability of lattice oxygen to suppress C2H4 overoxidation.
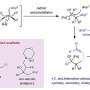
Moreover, this hybrid catalyst also accelerates the generation of Zn+–O– pairs to activate C−H bonds, stabilizes the reaction intermediate (*OCH3) to achieve the C−C coupling, and promotes the generation of low valence Zn to accelerate the dehydrogenation of the *OC2H5 into C2H4.
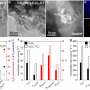
Therefore, a stable photocatalytic methane conversion performance can be achieved over C-ZnO/Au with a stoichiometric generation of ethylene and hydrogen.
"Due to the high dissociation energy of methane and the complex surface structure of catalysts, it is of great significance to correlate the structure of the active center with its reactivities. The structure of the C-ZnO/Au is resolved by density functional theory calculation, and the mechanism of highly selective conversion of methane to ethylene is revealed by in situ characterization," Xiao says.
A few implications thus emerge for designing photocatalysts for methane conversion: 1) improving the photogenerated charge carrier separation efficiency of catalyst to promote methane conversion; 2) promoting electron transfer to the antibonding orbitals of C−H bond to accelerate methane activation; 3) stabilizing the reaction intermediate to enhance the C−C coupling.
The study is published in the journal Science China Chemistry.
More information: Jing Wang et al, Photocatalytic nonoxidative coupling of methane to ethylene over carbon-doped ZnO/Au catalysts, Science China Chemistry (2023). DOI: 10.1007/s11426-023-1766-8
Provided by Science China Press