New molecule sets stage for nickel as a 'greener' photocatalyst, reveals key steps in reaction process
In recent years, the golden word in precious metals is palladium.
A crucial component in automobile catalytic converters and in emerging hydrogen fuel cell technology, the demand for this rare silvery white transition metal continues to outstrip supply, driving its price per ounce far above gold and silver.
Palladium and other rare, costly precious metals like platinum, iridium, and ruthenium, are also crucial in chemical transformations, specifically transition metal catalysis, which has become an indispensable tool for putting together complex molecules in the development of pharmaceutical drugs, polymers, and other useful chemicals.
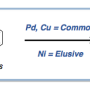
The scarcity and expense of these precious metals has created a need to develop catalysts from transition metals that are more abundant and generally cheaper, like nickel, a cousin of palladium.
As a result, the last decade has seen a dramatic expansion of new catalytic bond-forming transformations involving nickel.
"We know from the literature that nickel complexes are extremely useful in performing some transformations, maybe better than other transition metals out there," said Liviu Mirica, William H. and Janet G. Lycan Professor of Chemistry at the University of Illinois at Urbana-Champaign. "People have gotten very good at optimizing conditions for specific transformations, so we are slowly getting to where nickel could rival palladium in these transformations."
More recently, scientists have been focusing on developing nickel catalysts that can be directly photoinitiated by light, which Mirica said has proven to be a very successful area of research, producing reactions that have not been previously possible.
However, they still require the use of an additional photocatalyst—typically based on precious metals such as iridium or ruthenium, which are even more expensive than palladium.
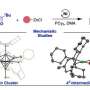
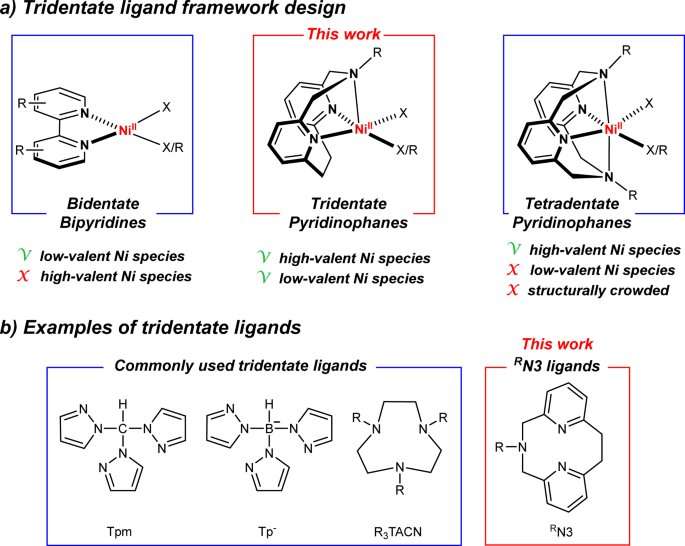
In a paper recently published in Nature Communications, Mirica and postdoctoral researcher Hanah Na report their work on the development of a completely novel tridentate ligand that coordinates with nickel to create a catalyst that can be directly activated by light to form a carbon-oxygen bond without the use of an additional photocatalyst. C-O bonds are prevalent in many natural products, pharmaceuticals, and agrochemicals.
Mirica and Na believe their new class of tridentate pyridinophane ligands (RN3) can lead to the development of new nickel catalysts and are a practical platform for detailed mechanical studies of other nickel-catalyzed chemical reactions.
"It is a competent catalyst and on top of it, it can actually do this photocatalysis by itself; it doesn't require these other photocatalysts," Mirica said. "It opens up many avenues of research that we think could be used for many additional applications."
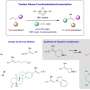
These tridentate pyridinophane ligands (RN3) build on previous work by Mirica, who had already developed a novel four-pronged molecule known as a tetradentate ligand, whose structure resembles the pocket of a baseball glove. This ligand structure promoted rapid C-C bond forming reactivity while also stabilizing the higher oxidation states of nickel.
"It's very stable. But all of those intermediates over the past decade have been way too stable. They're not competent in catalytic applications," Mirica said.
Then there is the bidentate ligand framework bipyridyl that most chemists are using in nickel photocatalytic processes, which provides enhanced reactivity and the ability to adjust optimization to get the desired reaction.
"It's great for catalytic chemistry but you can't isolate or see these special nickel species," Mirica said.
Typically, Mirica explained, classic organic chemists have a particular chemical transformation in mind and try whatever catalysts they think will be good, and whatever conditions or additives would be useful and optimize it, focusing on a very specific transformation.
"We have a slightly different approach: a metallo-centric approach, and in this case nickel is the metal of interest," he said. "I am interested in being able to design, isolate, characterize nickel complexes with different coordination numbers, different ligand environments, and in different oxidation states, which ultimately will dictate their reactivity."
This latest ligand structure is somewhere between the other two.
"We open up a coordination site, we open up that nickel center, by removing one of the four nitrogens, to allow other things to bind to it and eventually it allows you to do catalytic activity, but still be able to isolate and characterize intermediates," he said.
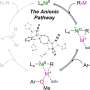
Their novel tridentate ligand enabled them to reveal for the first time the key reaction steps and intermediate species in this catalytic cycle. An in-depth mechanistic understanding of Ni-mediated photocatalysis is essential for rational reaction design and optimization of the nickel-mediated chemical process, the researchers explain in the report.
Their mechanistic study employed techniques including nuclear magnetic resonance (NMR), electron paramagnetic resonance (EPR), in situ infrared (IR) spectroscopy and electrochemical and photophysical measurements, and computational studies.
From a mechanical perspective, the photocatalytic cycle is well-understood, but the Ni-mediated redox cycle has remained a mystery. Paramagnetic Ni(I) and Ni(III) species are assumed to be part of the process, but have not been thoroughly investigated, and the key catalytic steps of oxidative addition, trans-metalation, and reductive elimination at the nickel centers have never been directly observed.
In the past several decades, Na explained, visible light-mediated photoredox catalysis has made vital contributions in the field of synthetic organic chemistry. Traditionally, developing new methodologies and reaction condition optimization are often achieved by trial and error rather than being based on a thorough understanding of the underlying reaction mechanism.
Na said this might be because understanding of the underlying chemistry requires a major contribution from the inorganic and organometallic chemistry fields (beyond the scope of the research interests in synthetic organic chemistry), including the synthesis and characterization of related metal complexes and study of their photochemistry and photophysics.
"As inorganic and organometallic chemists, we want to contribute to this emerging research field, mostly focusing on unraveling clues to understand underlying reaction mechanisms—which is not much done by organic chemists," Na said. "We believe that our work would provide crucial insight into the reaction design and search for new chemical transformations in the burgeoning field of photoredox catalysis, and thus can impact both the organic and inorganic chemistry community."
The goal, Mirica explained, is to unleash new reactivity that could ultimately be helpful to organic chemists, who could then employ this system and use it for very particular synthetic targets.
"They may not work now as well as the finely optimized or finely tuned systems that people use on a daily basis in an organic lab, but we hope that our new Ni catalysts will be commonly used several years down the line," Mirica said.
More information: Hanah Na et al, Deciphering the mechanism of the Ni-photocatalyzed C‒O cross-coupling reaction using a tridentate pyridinophane ligand, Nature Communications (2022). DOI: 10.1038/s41467-022-28948-8
Journal information: Nature Communications
Provided by University of Illinois at Urbana-Champaign